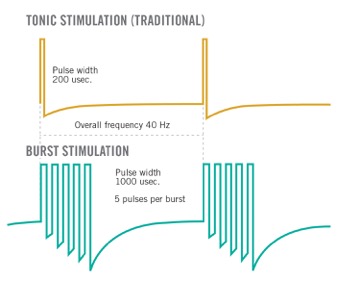
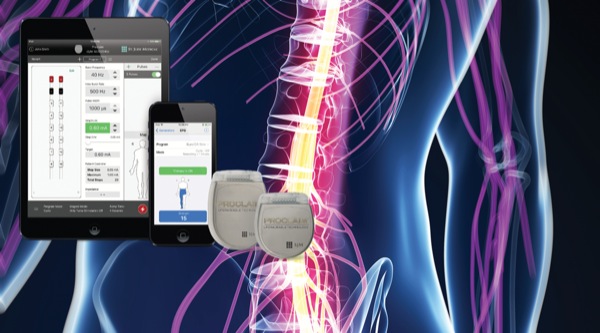
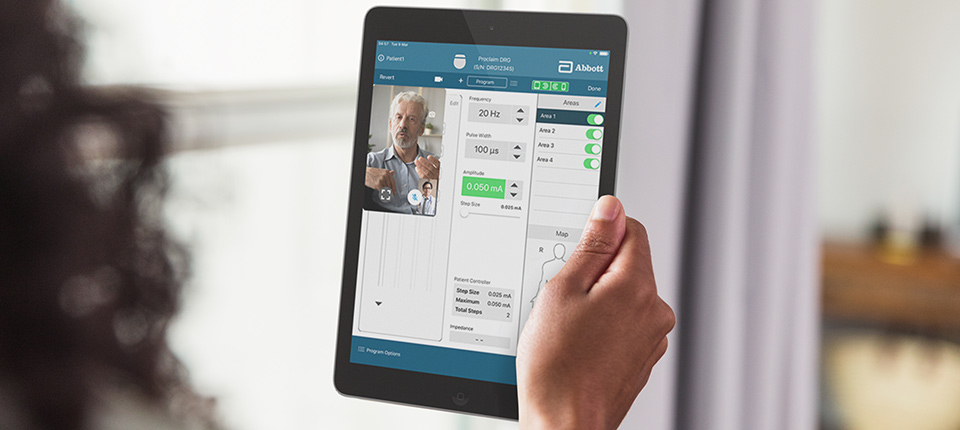
FDA Approves Abbott's "Low Dose," Recharge-Free Spinal Cord Stimulation System with up to Ten Year Battery Life* for People Living with Chronic Pain
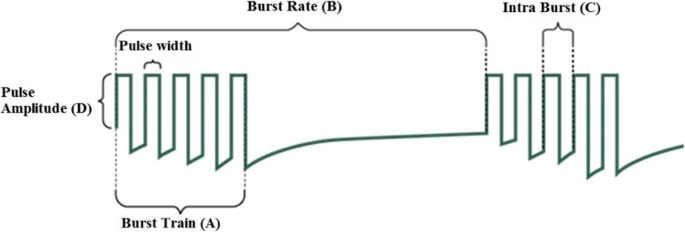
Multicentre, clinical trial of burst spinal cord stimulation for neck and upper limb pain NU-BURST: a trial protocol | Neurological Sciences

Spinal cord stimulation for the treatment of peripheral neuropathic pain | Tidsskrift for Den norske legeforening
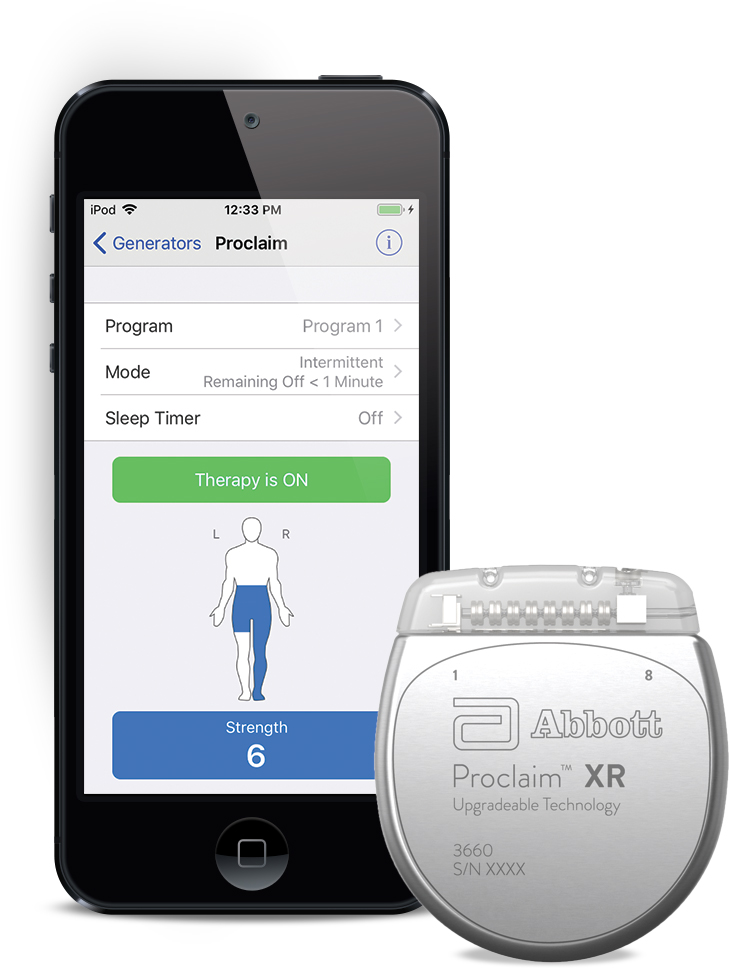
FDA Approves Abbott's "Low Dose," Recharge-Free Spinal Cord Stimulation System with up to Ten Year Battery Life* for People Living with Chronic Pain
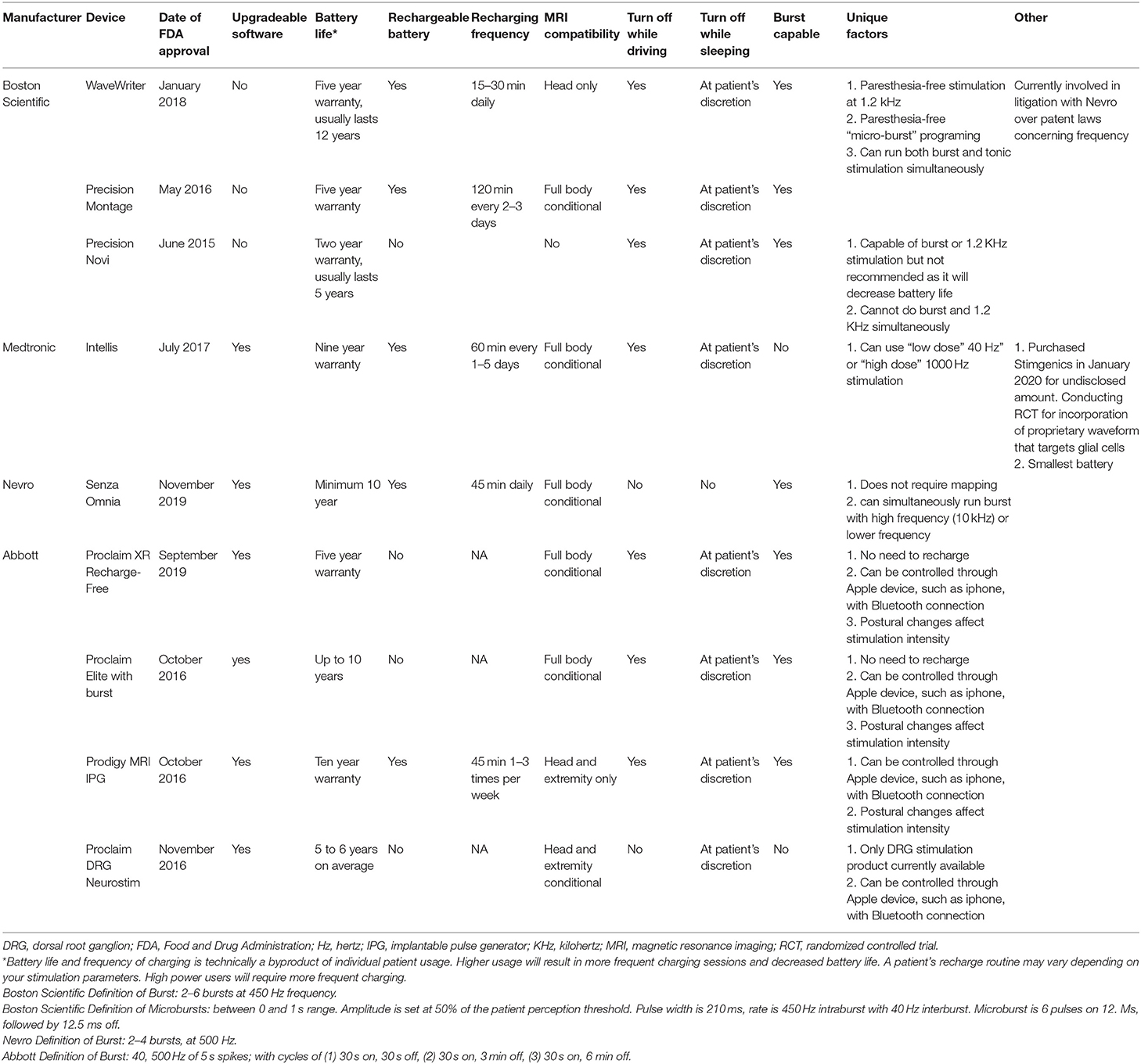
Frontiers | Survey of Spinal Cord Stimulation Hardware Currently Available for the Treatment of Chronic Pain in the United States

Proclaim™ XR Spinal Cord Stimulation (SCS) System with BurstDR™ Technology: Paddle Lead Implant Procedure | Intended to be viewed upon completion of a successful evaluation, this video illustrates the permanent implant procedure

Abbott launches Eterna spinal cord stimulation system for the treatment of chronic pain - Spinal News International